
Phosphorus pentachloride may be used in the synthesis of following compounds: benzenesulfonyl chloride malonic dinitrile aromatic sulfonyl chlorides Phosphorus pentachloride may be used as halogenating reagent to convert any substituted aldehyde into the corresponding vinyl halide. Up to 70 mg/m3: (APF = 2000) Any supplied-air respirator that has a full facepiece and is operated in a pressure-demand or other positive-pressure mode. (APF = 50) Any supplied-air respirator with a full facepiece. The three P-Cl bonds are in one plain held at equilateral triangle corners and the remaining two bonds lie axially to the plane of three bonds.Up to 50 mg/m3: (APF = 50) Any self-contained breathing apparatus with a full facepiece. It exhibits a trigonal bipyramidal geometrical shape. Phosphorus pentachloride is nonpolar in nature as it is symmetrical in shape. Little has been published on the analysis of these chemicals: POCl 3 and PCl 3 were analyzed by GC-MS as well as by direct injection MS,15 as early as 1990. 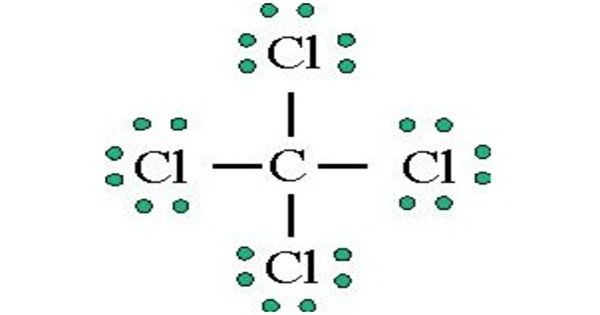
It is corrosive to metals and tissue.In Schedule 3, the three phosphorus containing inorganic chlorides phosphorus oxychloride (POCl 3), phosphorus tri-chloride (PCl 3), and phosphorus pentachloride (PCl 5) are listed. This heat may be sufficient to ignite surrounding combustible material. It is decomposed by water to form hydrochloric and phosphoric acid and heat. Phosphorus pentachloride is a greenish-yellow crystalline solid with an irritating odor. A process for the preparation of highly pure phosphorus pentachloride in the form of a free-flowing crystalline powder by reaction of phosphorus trichloride with chlorine, which comprises carrying out the reaction in two stages, phosphorus trichloride being combined with 0.1 to 0.999 times the molar amount of chlorine in the first stage at a temperature of 0 to 150° C, while stirring, 0 to. What is the equilibrium partial pressure of Cl 2 at 250 C. The equilibrium partial pressures of PCl 5 and PCl 3 are 0.875 atm and 0.463 atm, respectively. for the decomposition of phosphorus pentachloride (PCl 5) to phosphorus trichloride (PCl 3) and chlorine (Cl 2) PCl 5 (g) D PCl 3 (g) + Cl 2 (g) is found to be 1.05 at 250 C. 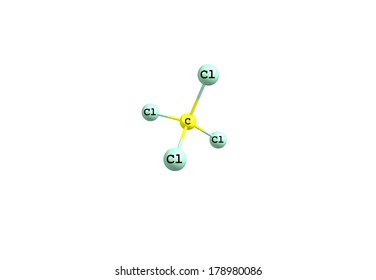
For the decomposition of phosphorus pentachloride (PCl 5) to phosphorus trichloride (PCl 3) and chlorine (Cl 2) PCl 5 (g) D PCl 3 (g) + Cl 2 (g) is found to be 1.05 at 250 C.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
